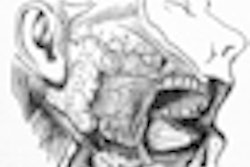
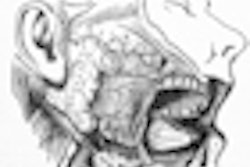
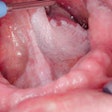
A University of Southern California (USC) study published in this month's Journal of the American Dental Association has found that the risk of osteonecrosis of the jaw (ONJ) in patients who have undergone oral bisphosphonate therapy could be higher than currently believed. Even short-term use can leave some patients vulnerable, according to the researchers.
“These results make a significant difference in the counseling and management of
patients on oral bisphosphonates.”
— Peter L. Jacobsen, Ph.D., D.D.S.
Most journals say the risk of osteonecrosis is negligible with oral bisphosphonates, but there is not enough data to back up this claim, said Parish Sedghizadeh, D.D.S., the director of the USC Center for Biofilms and one of the study authors, in a DrBicuspid.com interview.
"We found that approximately 4% of the patients who had a history of alendronate use developed osteonecrosis of the jaw," he said.
"These results make a significant difference in the counseling and management of patients on oral bisphosphonates," said Peter L. Jacobsen, Ph.D., D.D.S., the vice chairman of the ADA Council on Scientific Affairs. "The risk shown by this article is about two orders of magnitude higher than found in prior studies."
All healthcare providers and susceptible patients need to be made aware of the risks, he added.
But Merck, the maker of the oral bisphosphonate drug Fosamax, said in a written statement to DrBicuspid.com that the study is flawed, limited, and not a reliable source for valid scientific conclusions.
"Data from randomized, prospective clinical trials are the gold standard and are more reliable than data from studies like the one published in JADA," the company stated. "In controlled clinical trials involving more than 17,000 patients, contributing as much as 10 years' data with alendronate, there have been no reports of ONJ. This includes approximately 3,000 osteoporosis patients taking alendronate for 3 to 5 years and approximately 800 patients taking alendronate for 8 to 10 years."
The initial correlation between the use of oral alendronate and ONJ was not considered statistically significant by an expert panel appointed by the ADA Council on Scientific Affairs in 2006. The panel suggested then that routine dental treatment generally should not be modified solely on the basis of oral bisphosphonate therapy, the USC study authors noted.
Their recommendation was reaffirmed in 2008.
"We conducted a study to address the finding that in a patient population at the University of Southern California in Los Angeles, ONJ secondary to alendronate therapy is more common than suggested by the manufacturer and the ADA's expert panel," the authors wrote.
The study, which appears in the January issue of the Journal of the American Dental Association (January 2009, Vol. 140:1, pp. 61-66), was conducted at the USC School of Dentistry. It included 208 patients with a history of alendronate use, nine of whom had active ONJ and were being treated in USC clinics.
None of the patients was referred for evaluation or treatment of ONJ, excluding the possibility of referral bias, the authors noted.
Of the 208 patients, 66 underwent simple dental extraction without treatment modifications or preventive measures, and four developed ONJ at the extraction site postoperatively. Another five developed ONJ after denture-related mucosal ulceration.
The patients who developed bisphosphonate-associated osteonecrosis were all women between the ages of 63 to 80 who had received alendronate for osteoporosis for 12 months or longer.
Osteoporosis affects more than 10 million Americans, and alendronate is the most widely prescribed oral drug; it was the 21st most prescribed drug on the market in 2006, the authors noted. Despite these numbers, there are no epidemiologic data or research to support the risk of developing ONJ after its use, they added.
"Our data suggest that the risk of developing ONJ is much higher than initially reported," the authors noted.
This is not the first study of risk/incidence of ONJ in patients taking oral bisphosphonates, but it is the first to measure patients directly, explained Dr. Jacobsen.
The other studies estimated the risk from physicians voluntary reporting in the U.S. (which has a high risk of underreporting), physicians reporting in Australia (where such reporting is required, rather than voluntary, so the numbers are considered more accurate), or computerized analysis of insurance claims data with no patients or physician involvement, he added.
"If the current study holds up and future studies in other centers document its accuracy, then the prior study methods have appreciably underestimated the risk," Dr. Jacobsen emphasized.
Dental treatment implications
These findings clearly have implications for dentists working with patients who have a history of oral bisphosphonate use.
Alternate treatment options may be considered for unnecessary extractions, and good oral hygiene should be achieved before necessary extractions to minimize microbial load, according to the study authors.
They also recommend more routine and vigilant follow-up and use of chlorhexidine rinse preoperatively and postoperatively.
"Patients should get a dental consult before starting oral bisphosphonate therapy," Dr. Sedghizadeh said. "There should be comanagement of such patients by dentist and physicians, and it is important that they are involved in the informed consent process."
It is also important to identify patients who are at risk and focus on risk assessment and prevention protocols, he added.
"Most of the patients receiving alendronate at USC who developed ONJ did so after routine tooth extraction, suggesting that perhaps these patients should be identified as an at-risk population and preventive measures should be taken," the study authors noted.
There is a need for more studies in this area, Dr. Sedghizadeh said. People will say the risk may not be negligible if more studies like this come out, he added.
More science is needed to better understand why some patients get ONJ and others do not -- as well as what can be done to minimize the likelihood of this serious condition among susceptible patients, Dr. Jacobsen noted. This kind of patient-based data is what the ADA has said is needed all along, he emphasized.
Additional studies will be needed to better delineate the risk across the entire population of alendronate users, he concluded.