ActoGeniX has gained U.S. FDA clearance to conduct a phase IB clinical trial of a new treatment for oral mucositis in cancer patients, the company announced.
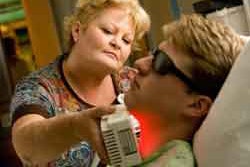
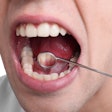
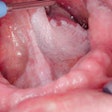
There is currently no effective cure for the severely painful and debilitating inflammation and ulceration of the mucous membranes lining the mouth that makes eating, drinking, and speaking difficult or impossible.
Oral mucositis can affect up to 100% of cancer patients undergoing high-dose chemotherapy.
The FDA approval permits ActoGeniX to initiate a phase IB clinical trial in six major U.S. oncology centers. The treatment, known as AGO13, could become the first approved therapy for oral mucositis in patients undergoing treatment of solid tumors or head/neck cancers, according to ActoGeniX.
Copyright © 2009 DrBicuspid.com