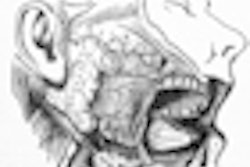
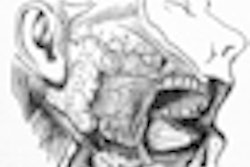
NEW YORK (Reuters Health), Jan 26 - Rituximab can improve saliva secretion and other parameters in patients with primary Sjögren's syndrome, researchers report.
Rituximab binds to the B cell surface antigen CD20 and could become the first causal systemic treatment for this disease, the investigators say.
At the University of Groningen, the Netherlands, Dr. J.M. Meijer and colleagues tested the drug in 30 patients with active primary Sjögren's syndrome. To be included in the trial, patients were required to have disease-related autoantibodies and a stimulated whole saliva secretion of at least 0.15 mL/min.
Earlier studies had shown that patients with at least some residual secretory ability might benefit most from rituximab, the authors explain in their paper, which appeared online January 13 in Arthritis and Rheumatism.
Twenty patients were randomized to receive IV rituximab, 1,000 mg, on days 1 and 15; the remaining 10 received placebo. On average, the patients were 43 years old, with a disease duration of 63 to 67 months.
For at least 6 to 9 months, rituximab-induced B cell depletion led to improvement in objective and subjective parameters, the researchers report. Compared to the placebo group, the rituximab group had significant improvements in the primary end point -- secretion of stimulated whole saliva (p = 0.004) -- and in various laboratory parameters (e.g., B cells and rheumatoid factor).
Rituximab-treated patients also had significant improvements in various subjective assessments, including multidimensional fatigue inventory (MFI), short-form 36 (SF-36), and visual analog scale scores. The rituximab group also had fewer extraglandular manifestations than control subjects.
One diabetic patient in the rituximab group developed a mild serum sicknesslike disease.
Most significant rituximab-associated improvements were observed between 12 and 36 weeks following treatment. In the placebo group, improvement of most variables occurred 5 weeks after the first infusion.
The authors theorize that improvements in the placebo group might have been related to prednisone given before and after the infusions, although they admit that data on the effect of prednisolone on Sjögren's syndrome symptoms "are inconclusive."
The research team concludes: "This study indicates that rituximab could be an effective and safe treatment ... for patients with primary Sjögren's syndrome."
Arthritis Rheum 2010.
Last Updated: 2010-01-25 15:32:44 -0400 (Reuters Health)
Copyright © 2010 Reuters Limited. All rights reserved. Republication or redistribution of Reuters content, including by framing or similar means, is expressly prohibited without the prior written consent of Reuters. Reuters shall not be liable for any errors or delays in the content, or for any actions taken in reliance thereon. Reuters and the Reuters sphere logo are registered trademarks and trademarks of the Reuters group of companies around the world.