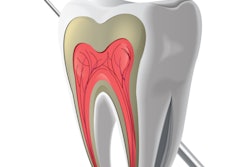
Glass ionomers (GIs) have officially been a part of dentistry since 1972. But for all practical purposes, they are a derivation of the earlier silicate cements. Alan D. Wilson, D.Sc., a chemist in the U.K., guided the conversion by transforming silicate cements into glass ionomers.
Since their introduction, glass ionomers have undergone many changes and have been endorsed for even more clinical applications. Today there are three different formulations of glass ionomer restorative materials:
- Traditional self-cured glass GIs
- Resin-modified GIs (RMGIs), sometimes called resin-reinforced GIs
- Compomers, also known as polyacid-modified composite resins
Traditional GIs
Traditional GIs are characterized by self-cured, acid-base setting reaction, chemical bonding to enamel and dentin, fluoride release, biocompatibility, and acceptable aesthetics. Generally, the major advantage of glass ionomers is their potential cariostatic effect due to the fluoride release and antibacterial activity. The GI releases fluoride when the surrounding area becomes an acidic (low pH) environment, as seen with the beginning process of carious activity.
Unlike adhesive resins, which bond mechanically to demineralized enamel and dentin, glass ionomers bond chemically to mineralized tooth structure through an ion exchange mechanism. Glass ionomers are formed through a combination of aqueous polyacrylic acid and finely powdered fluoroaluminosilicate glass. The glass is attacked by the acid, and silica-oxygen-aluminum bond hydrolysis leads to the release of cations that ionically cross-link the polycarboxylate chains. The ionic bond is created through chelation of the polyacids' carboxyl groups with the calcium in the apatite of the enamel and dentin.
When using this type of GI, the cavity surface must first be conditioned by applying a solution of polyacrylic acid, which is much less aggressive than phosphoric acid and is intended to remove the smear layer while increasing wettability. The bond strength to enamel is always greater than that to dentin because of the higher and more uniform inorganic content (Northwest Dentistry, November-December 2008, Vol. 87:6, pp. 13-19).
The mechanical properties of a GI are far lower than those of traditional composites. For instance, a typical micro/nano-hybrid universal composite will have a compressive strength of about 300 MPa and a tensile strength of at least 50 MPa, while GI's are about 150 MPa and 25 MPa, respectively. GIs also lack inherent toughness. That means that they cannot withstand high-stress concentrations. This is one reason they may not be the material of choice for pit-and-fissure sealants, plus they have much lower bond strengths compared to traditional pit-and-fissure resins (Anusavice KJ. Phillips' Science of Dental Materials, 11th ed., W.B. Saunders, 2003).
Traditional GIs are supplied as a powder:liquid and are self-cure, regardless of whether they are mixed by hand or with a mechanical device. Examples of this category:
- Ketac-Cem (3M ESPE)
- Fuji I (GC America)
Resin-modified GIs
"Resin-modified" specifically refers to the addition of polymerizable resin groups to the original GI formulation. This usually includes a methacrylate resin and HEMA (2-hydroxyethyl methacrylate). The result is a complex liquid that maintains acid reactivity independent of the newly acquired ability to be light polymerized.
It is important to recognize that the traditional acid/base setting reaction of the self-cure glass ionomer is still present and will continue as normal. Once polymerized, the resins will impart strength and also protection to the ongoing acid-base reaction from contamination, dehydration, and water sorption. Resin-modified GIs are thus occasionally referred to as "dual-cure"; the related term "tricure" emerged because some manufacturers also include a chemical initiator for the HEMA resin (Journal of the California Dental Association [JCDA], June 2003, Vol. 31:6, pp. 483-492).
3M was the first company to market RMGIs. The company's Vitrebond product, the first RMGI, is a liner that was introduced as a restorative RMGI in 1988, according to Afshin Falsafi, Ph.D., Ch.E., senior product development specialist with 3M ESPE.
Examples of low-viscosity/cement-type RMGIs include:
- Rely X Luting and Rely X Luting Plus (3M ESPE)
- Fuji Plus (GC America)
Examples of higher viscosity restorative RMGIs include:
- Fuji Filling LC (GC America)
- Ketac Nano (3M ESPE)
 Figure 1: The Ketac Nano delivery system employs a swiveling, static mixing tip, eliminating the need for trituration.
Figure 1: The Ketac Nano delivery system employs a swiveling, static mixing tip, eliminating the need for trituration.
The RMGI group also improved on the traditional GIs by increasing available shade selection and improving polishability, but these properties are still far below those of universal composites. To overcome this deficiency, a few years ago 3M introduced a nano-filled RMGI, the Ketac Nano (known as Ketac N100 outside the U.S.). This product incorporates nanotechnology from the company's flagship composite, Filtek Supreme.
A clinical evaluation of Ketac Nano in the October 2007 Clinicians Report cited enhanced polishability, reduced surface roughness, and improved surface gloss for Ketac Nano compared to other RMGI restoratives. Another feature of Ketac Nano is that it is supplied with a no-rinse acidic primer, unlike other GI/RMGIs that require placing polyacrylic acid, then rinsing it off. This product offers the dentist two delivery options:
- A dual-barrel paste:paste system for easier and more predictable hand mixing than powder:liquid products.
- Quick Mix capsules that require no trituration. This is identical to the delivery of impression materials in which the mixing tip contains a static mixer inside the barrel.
Compomers
Compomers, originally introduced in Europe, have been available since 1993. Compomers combine components and properties of both composites and GIs. Unlike true GIs, however, compomers are resin-based materials that do not contain water. In addition, the polymerization of compomer restoratives does not involve an acid-base reaction.
Compomers are actually light-polymerized composite resin restoratives, modified to contain ion-leachable glass particles and freeze-dried polyalkenoic acid. In the absence of water, the compomer composition prevents the glass particles and acid from reacting. Water uptake in the oral cavity, however initiates an acid-base reaction between the glass and anhydrous acid, resulting in the diffusion of low levels of fluoride ion from the restoration. Numerous in vitro studies have shown these fluoride release levels to be significantly lower than those measured for conventional and RMIs, according to the June 2003 JCDA article.
 Figure 2: Comparing fluoride release of various restoratives.
Figure 2: Comparing fluoride release of various restoratives.
Mechanical properties of compomers tend to be somewhat inferior to those of conventional composite resins, but greater than GIs and RMGIs. By adding resin to a traditional GI, all mechanical properties are significantly improved but fluoride release is compromised. However, the RMGIs and compomers experience very little erosion compared to traditional GIs.
The compomers are packaged in a single component syringe or single dose, like universal composites, and are light-cured. Examples would be:
- F2000 (3M ESPE)
- Dyract (Dentsply)
The greatest release of fluoride from any of these materials is highest early on, then it diminishes over time. A traditional GI releases more fluoride than RMGI, which releases more than compomers. Daily exposure to fluoride, such as toothpaste, has been shown to provide some recharging of the fluoride (Compendium of Continuing Education in Dentistry, March 2008, Vol. 29:2, pp. 82-91).
Indications
The traditional use of any GI product has been in the pediatric field, taking advantage of the cariostatic properties in children with compromised dentition or poor home care. However, as our population ages, we are now seeing more use of the GIs in geriatrics. Due to the increase in the use of various medications and higher incidence of cancer treatments, xerostomia is becoming more common in the 60 and older age group. Saliva normally bathes and rinses off our teeth, which helps prevent caries from starting. Xerostomia increases the probability of root caries, and oftentimes they occur where there was no previous restoration.
The original GIs only served as cements and liners/bases. Today, given that we have three choices of GI-related restoratives, the clinical indications have multiplied to include:
- Cement
- Liner/base
- Restorations -- especially pediatric and geriatric uses
- Core buildups
- Tunnel restorations
- Sandwich restoration -- open and closed technique
- Retrograde filling material
- Pit-and-fissure sealants
- Root caries or cervical restorations
- Temporary or A.R.T. restorations
- Endodontic sealer
It is not advisable to alter the powder:liquid ratio of a GI to change its intended viscosity because this also alters the acid:base reaction. With the expanded use of the GI family, manufacturers have given us choices with various viscosities to suit our needs. For cementing, or bases and liners, there are lower viscosity formulations, while the restoratives are available in medium-high viscosity for ease of placement, such as Fuji IX GP and Ketac Nano.
As shown in this article, the GI family has evolved to give dental practitioners many bioactive alternatives to traditional restoratives.
Copyright © 2010 DrBicuspid.com