The U.S. Food and Drug Administration granted 510(k) clearance to dental imaging software from dental practice management software developer Oryx Dental.
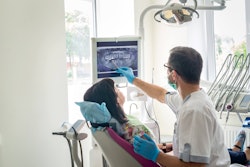
The company can now sell its Oryx Cloud imaging software directly to dental practices. The software acquires, views, annotates, and organizes dental x-rays and color images. It integrates with most dental imaging devices, including intraoral x-ray sensors, intraoral cameras, and scanners.
The software's open integration aims to allow clinicians to mix and match acquisition devices from a variety of manufacturers or consolidate cost on its dental management software, according to Oryx. Oryx's software can be used as a standalone product that integrates into existing systems, or it can be paired with Oryx's dental practice management software.