The HPV vaccine could eliminate recurrent oral squamous cell papillomas, according to a new case report and literature review in JAMA Dermatology. An immunocompetent patient with chronic verrucous papules was cleared of all lesions after receiving the quadrivalent HPV vaccine.
 Nika Cyrus, MD.
Nika Cyrus, MD."Currently, the HPV vaccine is not commonly utilized for treatment of cutaneous verruca vulgaris or oral warts (squamous cell papillomas)," lead researcher Nika Cyrus, MD, told DrBicuspid.com. "Our review of the literature revealed eight similar cases of resistant, even disseminated warts, which were successfully treated with the HPV vaccine." Dr. Cyrus is a resident at the University of Texas Southwestern Medical Center in Dallas.
HPV is the underlying cause of mucocutaneous verruca vulgaris and squamous cell papillomas, also known as oral warts. Topical and destructive treatments are particularly challenging in patients with disseminated disease, the study authors noted.
"The HPV vaccine should be mandatory for adolescents," researcher John J. Stern, MD, told DrBicuspid.com. He is the chief of the division of infectious diseases at Pennsylvania Hospital, University of Pennsylvania Health System in Philadelphia.
New treatment for oral warts?
 John J. Stern, MD. Image courtesy of Pennsylvania Hospital.
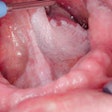
John J. Stern, MD. Image courtesy of Pennsylvania Hospital.In this case report, U.S. researchers documented a healthy, immunocompetent white man in his 60s with recurrent mucocutaneous lesions on his lips, tongue, and buccal mucosae. Three months after receiving the quadrivalent HPV vaccine, he had complete resolution of all papillomas and had no recurrence of lesions at 18 months, one year after receiving the last dose of vaccination (JAMA Dermatol, September 2, 2015).
"We treated our patient with multiple, recalcitrant oral warts with the HPV vaccine, which surprisingly resulted in complete clearance of the lesions after three months," Dr. Cyrus noted. "It is possible that the HPV vaccine can activate the immune system against nonvaccine HPV types resulting in a therapeutic response. Randomized clinical trials are necessary to further investigate the efficacy of the HPV vaccine for treatment of oral and cutaneous warts."
HPV-related diseases
Earlier studies show that noncervical HPV-related diseases cost billions of dollars annually, and the number of HPV-related oropharyngeal cancer cases is predicted to exceed cervical cancers by 2020.
A 2014 Centers for Disease Control and Prevention (CDC) study found that currently available HPV vaccines could prevent most oropharyngeal cancers in the U.S. (Emerging Infectious Diseases, May 2014, Vol. 20:5, pp. 822-828). Currently, there are about 85,000 cases worldwide of oropharyngeal cancers annually. In the U.S., about 12,000 new cases are diagnosed annually, and most are classified histologically as oropharyngeal squamous cell carcinoma.
“The HPV vaccine should be mandatory for adolescents.”
In the current report, the patient first noted the appearance of the lesions 18 months before presentation and had undergone multiple surgical excisions of the lesions by an otorhinolaryngologist with subsequent recurrences. His sexual history was significant for oral sex with multiple male partners, and he smoked a half pack of cigarettes daily.
The patient tested negative for hepatitis and HIV. A biopsy indicated the patient had acute and chronic inflammation; he was diagnosed with squamous cell papilloma with HPV viral effect.
Within four weeks after receiving the first dose of quadrivalent HPV vaccine, the patient reported marked improvement of the papillomas, the authors reported. He subsequently received the second and third injections of the vaccine at two and six months, respectively.
At the three-month follow-up, one month after the second dose of the vaccine, the patient had complete resolution of all papillomas. At the six-month follow-up after initial presentation, the patient still had complete resolution of all mucocutaneous papillomas with no recurrence.
How the body clears HPV
Cell-mediated immunity and cytotoxic T lymphocytes play an important role in the body's ability to clear HPV-infected cells, the authors noted. In the case of HPV vaccination, protection is also achieved via the induction of humoral immune responses, such as the production of HPV antibodies.
In this case, the presence of HPV type 32 within the lesional mucosa using laser microdissection polymerase chain reaction (PCR) suggests the possibility of cross-protection against nonvaccine HPV types after administration of the quadrivalent vaccine.
The possible mechanism for resolution of warts after quadrivalent HPV vaccination includes presentation of the L1 protein antigen, which is delivered systemically by antigen-presenting cells to T and B cells in lymph nodes, leading to production of HPV-specific CD4 helper T cells, cytotoxic T cells, and immunoglobulin-producing B cells, according to the authors.
More trials needed
Randomized clinical trials are needed to assess the efficacy of the quadrivalent HPV vaccine to treat cutaneous and mucosal warts, especially in cases of recalcitrant or disseminated lesions, the authors wrote. In future trials, HPV types could be determined, and the production of specific antibodies and T cells against HPV types in cutaneous and mucosal warts could be monitored to delineate underlying mechanisms of therapeutic response, they concluded.
"The vaccine should be offered to all gay men who are not in a monogamous relationship, despite the fact that insurance won't pay for it, with the idea of minimizing cancer in this at-risk population," Dr. Stern said.
"It is effective for prevention of cervical and anal cancer, as well as oral and genital warts," Dr. Cyrus noted.