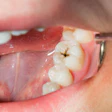
A surgically implanted device that moves a person's tongue forward during sleep reduces sleep apnea severity in children with Down syndrome, according to a study published April 21 in JAMA Otolaryngology -- Head & Neck Surgery.
The pacemakerlike device, called an upper airway hypoglossal stimulator, sends an electrical pulse to the tongue, causing it to contract when a person tries to take a breath. It has been used to effectively treat obstructive sleep apnea in adults, but this is the first phase I clinical trial to evaluate its use in children with Down syndrome -- a cohort more likely to experience sleep apnea and less likely to respond to traditional treatments.
"This study was born out of the frustration of not having an effective treatment option for children with Down Syndrome who struggle with sleep apnea," senior study author Dr. Christopher Hartnick, director of the division of pediatric otolaryngology and the pediatric airway, voice, and swallowing center at Mass Eye and Ear and professor of otolaryngology, head and neck surgery, at Harvard Medical School, said in a statement.
Need for new treatment options
Obstructive sleep apnea, or OSA, occurs when the airway is blocked during sleeping. The condition can result in restless sleep, snoring, and difficulty breathing, and children with sleep apnea report daytime sleepiness, learning challenges, and behavioral problems.
While the condition only occurs in 5% of children in the general population, up to 80% of children with Down syndrome have obstructive sleep apnea. Children with Down syndrome are more likely to have a larger tongue, which may predispose them to the condition.
What's more, children with Down syndrome are less likely to benefit from or adhere to traditional treatment options for sleep apnea. Fewer than 30% of these children benefit from removing the adenoids and tonsils, and sensory difficulties prevent many from tolerating continuous positive airway pressure (CPAP) machines.
"Sleep apnea significantly impacts these children, often affecting their language, behavior, and ability to do well in school. Any diminishment in these areas is critical for families, and our study suggests we may finally have an option to help," Hartnick said.
Trial shows promising improvements
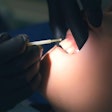
The new multicenter prospective trial found the pacemakerlike device can be safely implanted and successfully reduce the number of apnea events in children with Down syndrome and severe obstructive sleep apnea.
The study included 42 patients, ages 10 to 22, who underwent surgery for implantation of the upper airway stimulation device. The device was turned on one month after the surgery, and patients were followed for one year.
At the one-year mark, most patients responded well to the treatment, with two-thirds experiencing a 50% decrease in apnea-hypopnea index (AHI). On average, the AHI dropped by nearly 13 events per hour.
Participants also reported improved quality of life. Nearly 78% of patients had a moderate or large improvement on an obstructive sleep apnea survey, and sleepiness scores improved by an average of 5 points on the 24-point Epworth Sleepiness Scale,
More so, the authors deemed the implant safe. The most common complication, temporary tongue or oral discomfort or pain, occurred in five patients and resolved in a few weeks.
However, one patient experienced worsening central apnea at the one-month mark, and four patients needed to be readmitted to the hospital, often for complications related to picking at the incision site.
More research on the way
There's still questions around the long-term success of the implant in children with Down syndrome. The upper airway stimulator battery is expected to last about 10 years, and patients will require a surgical procedure to replace both the battery and generator.
Additionally, the study doesn't answer which children with Down syndrome are the best candidates for an implant, since some children did not experience improvements or two did not use the device for at least four hours per night.
The study also questions whether AHI is the best metric for evaluating sleep apnea in this population. AHI scores that count as severe are lower for children than they are for adults, and many of the children had AHI scores that indicate severe sleep apnea even after device implantation.
"Although our results were compelling, it revealed that we need to carefully look at what other outcome measure we're using," he stated. "That's what our next study is looking for."
Future studies need to account for some of the limitations in this trial, including lack of a control group and the relatively small sample size. Meanwhile, Hartnick and colleague Dr. Brian Skotko, MPP, are already working to refine how to measure outcomes for this population.
Using a $4 million, five-year U.S. National Institutes of Health grant, the duo will investigate whether the implant can improve neurocognition and language in children with Down syndrome.
"When parents reported anecdotally that the implant seemed to improve speech in their loved ones, we knew that we needed to investigate this further," said Skotko, the Emma Campbell endowed chair on Down syndrome at Massachusetts General Hospital, in a statement. "The NIH grant will enable us to formally analyze improvements that the implant may or may not have on speech and cognition."