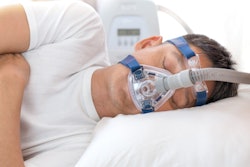
The U.S. Food and Drug Administration (FDA) is warning that Philips Respironics continuous positive airway pressure (CPAP) machines, which are used to treat obstructive sleep apnea, should be monitored for signs of overheating following reports of fire and burns.
The agency issued the advisory after noticing an increase of medical device reports associated with DreamStation 2 CPAP machines from Philips Respironics, a U.S. subsidiary of Royal Philips. Between August 1, 2023, and November 15, 2023, the agency saw a sharp increase of more than 270 reports of thermal issues associated with the machines. Prior to this, the agency had received fewer than 30 reports related to overheating issues since the machines were cleared for the market on July 10, 2020, according to an FDA press release dated November 28.
The report filed most recently cited issues such as burns and injuries to users, as well as fire, smoke, and other signs of overheating, while using the machine, according to the FDA.
“The FDA is committed to assuring this new safety issue is resolved expeditiously. We will continue to monitor the company’s handling of this, among other safety issues, to ensure they take appropriate steps to mitigate the risk to patients,” Dr. Jeff Shuren, director of the FDA’s Center for Devices and Radiological Health, said in the press release.
While the FDA is in ongoing discussions with the company about strategies to mitigate this safety problem, it recommends that those who use the machines should adhere to the manufacturer’s instructions and check the devices for unusual smells and appearance changes before and after use, according to the release.
Over the last three months, Philips Respironics filed the reports following a retrospective review of possible thermal complaints related to the DreamStation 2 since it was brought to market, according to a statement dated November 29 from Philips.
“The devices can continue to be used provided that the safety instructions in the instructions for use for the DreamStation 2 sleep therapy device are followed,” according to a statement.
This latest safety issue comes as the agency continues to oversee Philips’ handling of the June 2021 recall of the company's sleep therapy and respirator machines, including its CPAP machines. The machines that were recalled contained polyester-based polyurethane (PE-PUR) foam, which reduce sound and vibration but could break down and could potentially be breathed in or swallowed, which pose potential risks to those using the devices. However, the DreamStation 2 machines are not made with PE-PUR foam. These machines are made with a silicone-based foam and were not included in the June 2021 recall.
Based on the currently available evidence, the FDA does not believe that the foam in the machine is what is causing the overheating problems with the DreamStation 2 machines.
“This is a developing situation, and to date, reports gathered and analyzed by the FDA indicate the thermal issues may be related to an electrical and/or a mechanical malfunction of the machine, which may cause it to overheat in certain situations,” according to the release.