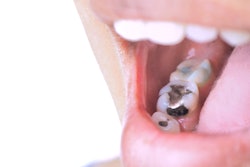
The FDA this week settled a lawsuit that sought to have mercury fillings banned from the U.S. market. While not agreeing to a ban, the agency has agreed to add warnings regarding the use of amalgams in some patient groups, and to complete a review that could lead to more stringent regulation of the substance.
"Dental amalgams contain mercury, which may have neurotoxic effects on the nervous systems of developing children and fetuses," the agency now states on its Web site. "Pregnant women and persons who may have a health condition that makes them more sensitive to mercury exposure, including individuals with existing high levels of mercury bioburden, should not avoid seeking dental care, but should discuss options with their health practitioner."
As part of the settlement with Moms Against Mercury, Consumers for Dental Choice, and the other plaintiffs who filed the lawsuit against the FDA last December, the agency is now required to formally classify dental amalgam by July 2009, according to Peper Long, FDA spokesperson. In its 2002 proposed rule on dental amalgam, the agency proposed to classify it as a Category II device.
"Now that we are going to put this into a class, we will be able to put regulations in place that are more specific to dental amalgam," she said.
The agency has struggled for years to reach a definitive conclusion about amalgam fillings. In 2004 an FDA panel determined that there was no evidence of risk from dental amalgam -- even to pregnant women. But a 2006 committee disagreed. Even so, prior to the settlement of this lawsuit, the language on the FDA Web site regarding the safety of dental amalgams was less direct, noting only that "the possibility that dental amalgam could pose health risks that are not yet scientifically known." In addition, regarding potential health risks of mercury for pregnant women, the Web site previously stated: "The recent advisory panel believed that there was not enough information to answer this question."
In a letter posted June 3 on the Consumers for Dental Choice Web site, Charles G. Brown, an attorney for the consumer group, declared, "We have won our ten-year battle to get the Food and Drug Administration to comply with the law and set a date to classify mercury amalgam. The impact of the re-writing of its position on amalgam can hardly be understated."
The ADA said in a formal statement issued June 4 that the FDA settlement with Moms Against Mercury "in no way changes the federal agency's approach to or position on dental amalgam. The 'settlement' only affirms that the FDA will do what it has intended doing all along. In the settlement, the FDA agrees to complete the classification process of dental amalgam by July 28, 2009. The ADA has supported the proposed reclassification since the agency first proposed it in 2002 and continues to support it."
Currently, the FDA has different classifications for encapsulated amalgam and its component parts, dental mercury and amalgam alloy. "The ADA agrees with the pending proposal from the FDA to place encapsulated amalgam and its components under one classification. Based on extensive studies and scientific reviews of dental amalgam by government and independent organizations worldwide, the ADA believes that it remains a valuable, viable, and safe choice for dental patients," the organization said in its statement.
In addition to Mothers Against Mercury, plaintiffs in the lawsuit included four nonprofit groups, two public officials, three dental professionals, and two consumer advocates that Brown refers to as "victims."