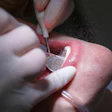
The debate over the regulation -- or lack thereof -- of teeth-whitening products heated up last month when the ADA petitioned the FDA to begin classifying bleaching chemicals.
The move stems from a 2008 ADA House of Delegates resolution seeking to petition the FDA to properly classify these chemicals. The resolution came amidst concerns about safety issues regarding unlicensed providers working in teeth-whitening businesses.
Teeth-whitening kiosks and centers run by nondentists have popped up across the U.S. and in other countries, attracting attention because they offer a less expensive alternative to having the work done by a dentist. Procedures that might cost $400 to $500 at a dentist's office typically cost only about $100 to $140 at these centers in the U.S.
“We understand … that the petition was not intended to apply to reputable OTC tooth whiteners.”
— Alissa Hammond, external relations manager, Crest Whitestrips.
But patient safety remains an issue, according to the ADA. Even so, the owners of the teeth-whitening centers insist they are not doing dentistry. They say they are only giving whitening agents to customers with instructions on how to perform the procedure themselves.
Nevertheless, several states -- including Tennessee, Oklahoma, Georgia, and Alabama -- have cracked down on these centers and tried to ban them from operating. Outside the U.S., Australia has taken a strong position against non-regulated facilities as well.
"The tremendous expansion of products available directly to consumers and application of products in venues such as shopping malls, cruise ships, and salons is troubling since consumers have little or no assurance regarding the safety of product ingredients, doses,or the professional qualifications of individuals employed in these non-dental settings," said ADA President Ron Tankersley, D.D.S., and Executive Director Kathleen O'Loughlin, D.D.S., in a letter sent November 20 to the FDA.
Too many unknowns
The FDA currently supports the use of hydrogen peroxide and carbamide peroxide as oral antiseptic agents, but extracoronal whitening/bleaching products have not yet been classified.
In its petition, the ADA raised the following concerns:
--Peroxides are irritants and potentially caustic to soft tissues.
--Tooth-whitening formulations continue to change at a rapid pace without specific scientific or regulatory guidance on safety. Bleaching agent type and concentration, formulation pH, application methodology, etc., have potential safety implications.
--Direct-to-consumer products may be easily over-used and abused by individuals who assume that the products are safe since they are readily available.
The petition states that the use of tooth whitening/bleaching materials is not risk-free and that oral soft tissues, tooth structure, and dental restorations can all be adversely affected by these products. Also, discolored teeth may be the only visible sign of pathology that requires treatment.
Daniel Meyer, D.D.S., ADA senior vice president of science/professional affairs, confirmed that the ADA is asking for this change out of concern for patients because of the increasing trend of teeth-whitening services being provided by unlicensed individuals in malls, kiosks, salons, ships, and other non-medical environments. "We don't know the ingredients, strength, or frequency of chemicals they are using," he told DrBicuspid.com. "These products need to be identified and regulated. There have been reports of people being harmed by these types of products and procedures." Also, not everyone is a candidate for teeth-whitening procedures, he noted. Staining on teeth could be from a variety of conditions such as developmental defects, trauma, infection, decay, necrotic pulps, discolored restorations, or stains from medications such as antibiotics. In addition, individuals may have other problems such as cracked teeth or restorations, gingivitis, periodontitis, oral lesions, or other pre-existing conditions that should be evaluated by a dentist before bleaching is considered. "Non-dentists are not educated or trained to diagnose these conditions," said Dr. Meyer.
Over-the-counter products
And what about over-the-counter (OTC) products? "With any whitening products, whether applied in unlicensed salons, self applied or professionally applied, we recommend that people first find out why their teeth are discolored and understand the source of the problem," explained Dr. Meyer. "It is beneficial to have a dentist do an examination to determine if bleaching is appropriate and will be effective before putting any kind of chemical on hard or soft tissue."
Alissa Hammond, external relations manager for Crest Whitestrips, told DrBicuspid.com that the company extensively tested its Whitestrips products to ensure that they are safe on enamel when used as directed.
"We understand from the ADA that the petition was not intended to apply to reputable OTC tooth whiteners such as Crest Whitestrips," she added. "The FDA specifically allows the sale and purchase of cosmetic products without the need for prior consultation with a doctor or dentist. Crest Whitestrips have been marketed as cosmetic products for years."
In 1992 the FDA agreed to evaluate the status of tooth-bleaching preparations to determine whether and how they should be regulated. But that evaluation was not completed, according to the ADA petition.
When asked for comment, the FDA's Center for Drug Evaluation and Research provided the following statement:
"FDA is reviewing the issues raised in this petition, and we can't comment at this time. We will issue our response when our review is complete."