Salivary mucins, key components of mucus, actively protect teeth from one of the principal bacteria that causes caries, Streptococcus mutans, according to a new study in Applied and Environmental Microbiology (October 24, 2014). The findings suggest that bolstering native defenses might be a better way to fight dental caries than relying on exogenous materials such as sealants and fluoride treatment, according to the study authors.
S. mutans attaches to teeth using sticky polymers that it produces, eventually forming a biofilm, a protected surface-associated bacterial community that is encased in secreted materials, according to lead author Erica Shapiro Frenkel, DDS, of Harvard University. As S. mutans grows in the biofilm, it produces organic acids as metabolic byproducts that dissolve tooth enamel, which is the direct cause of caries.

"When you use antibiotics or in certain hygiene measures, you kill a population of cells. But the problem with that -- for example, in your mouth or your digestive tract -- is that many of these cells are good for you and help protect you from other pathogenic events, interactions, and microbes," principal investigator Katharina Ribbeck, PhD, a biochemist and assistant professor in the department of biological engineering at the Massachusetts Institute of Technology (MIT), told DrBicuspid.com. "So ideally you want to maintain your microbiota, the diversity, while preventing virulence."
Having a large diversity of microbes on the body is highly beneficial, because these microbes bring desirable traits, she noted. For example, they help digest food in the oral cavity and might also have an immune protective function. But some microbes are opportunistic pathogens.
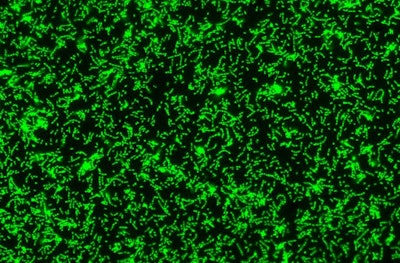
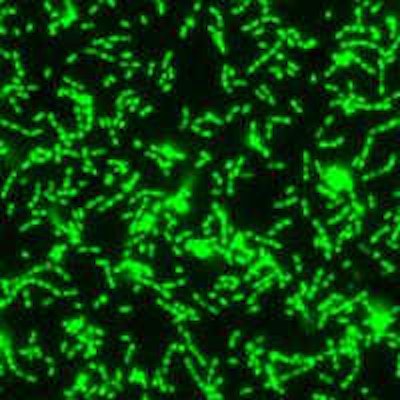
A fluorescent microscopy image of a S. mutans biofilm. Image courtesy of Nicole Kavanaugh of MIT and the American Society for Microbiology.
"The big question is how can you build a habitat, an eco-niche -- which is in the mouth in the mucosa and generally on wet surfaces of the body -- how can you build an eco-niche which allows the good and beneficial microbes to thrive while keeping the potentially harmful microbes from doing damage to the body?" Ribbeck said. "And with the mucins, we find they very elegantly manage to do so by preventing damage by microbes, by preventing them from attaching to surfaces."
For their study, the researchers used a hydroxy appetite as a proxy for tooth surfaces in the lab model.
The starting point for dental caries is not the presence of S. mutans but the bacterium's firm attachment with tooth surfaces, Ribbeck explained.
"Once these microbes attach themselves to the surface of teeth, they grow in this location with a matrix, and once they form this community, this group of bacteria, they become really difficult to remove," she said.
They qualify as a biofilm, forming cells that are connected to each other by an extracellular matrix material that they secrete and surround themselves with.
"Once they are in this biofilm state attached to a tooth surface, then they become really difficult to remove, not only with a toothbrush but they also become resistant to clearance with mouthwashes," Ribbeck explained.


The viscoelastic properties of mucus and saliva are attributed to mucins, large glycoproteins that play a key role in host defense and maintaining a healthy microbiome. Human salivary mucins protect teeth from dental cavities, which are primarily caused by S. mutans colonization.
"We found that salivary mucins don't alter S. mutans' growth or lead to bacterial killing over 24 hours. Instead, they limit biofilm formation by keeping S. mutans suspended in the liquid medium," stated Dr. Frenkel, who is also a doctoral student in Ribbeck's biological engineering lab at MIT, in a press release. "This is particularly significant for S. mutans, because it only causes caries when it is attached or in a biofilm on the tooth's surface."
So it is this biofilm's attachment to teeth, not so much the presence of microbes, that causes caries.
"When individual microbes form, they don't do that much dire harm to the body," Ribbeck noted. "Typically, it's only when they team up and build large groups that are associated to tooth surfaces, this is when they start to become problematic."
Dr. Frenkel pointed out that the oral microbiome is better preserved when naturally occurring species aren't killed. "The ideal situation is to simply attenuate bacterial virulence," she stated.
The study grew out of previous work in their lab showing that other types of mucins, such as porcine gastric mucins, had protective effects against common lung pathogens. With this in mind, the researchers suspected that salivary mucins would play a protective role, but they were unsure what that would be.
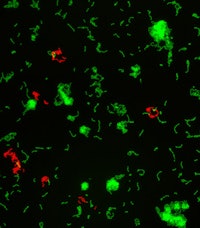
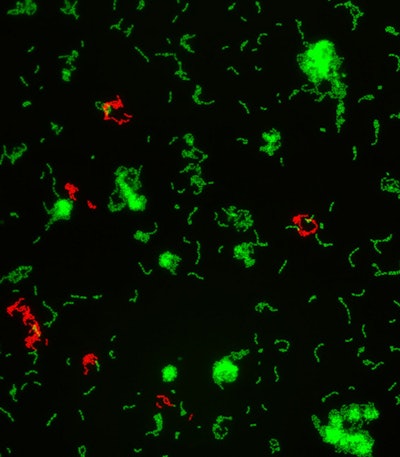
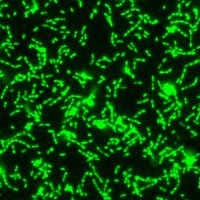
Two overlayed fluorescent microscopy images of S. mutans, the primary bacteria responsible for dental cavities. When S. mutans forms biofilms on the tooth surface, it produces organic acids as metabolic byproducts that dissolve tooth enamel. The green fluorescent image shows S. mutans biofilm mass when grown in the presence of sucrose, a substrate necessary for substantial bacterial attachment and biofilm formation. The red fluorescent image overlayed depicts S. mutans biofilm mass in the presence of sucrose and purified human salivary mucins. Mucins are key structural components of mucus and saliva that play an important role in host defense. Image courtesy of Nicole Kavanaugh of MIT and the American Society for Microbiology.
"Defects in mucin production have been linked to common diseases such as asthma, cystic fibrosis, and ulcerative colitis," Dr. Frenkel explained. "There is increasing evidence that mucins aren't just part of the mucus for structure or physical protection, but that they play an active role in protecting the host from pathogens and maintaining a healthy microbial environment. We wanted to apply these emerging ideas to a disease model that is a widespread, global public health problem -- caries. We chose to study the interaction of MUC5B [salivary mucins] with S. mutans because it is the primary caries-causing bacteria in the oral cavity."
Caries and periodontal disease are often caused by a decrease in normal saliva production. The research could be used to help scleroderma patients, who commonly have dry membranes of the mouth and eye. These symptoms are thought be caused by a dysfunction of tear or salivary glands secondary to an autoimmune process or tissue fibrosis.
The research may also help those who don't produce enough mucins in their saliva -- for example, in cases of dry mouth.
"We hope to find ways to replenish mucins, to reconstitute the natural protective system," Ribbeck said. "But also for just boosting your immune protection, your ability to prevent S. mutans and probably other microbes from causing damage."
Ribbeck's lab has started building synthetic versions and engineering mucin versions that could be used in existing hygiene products and even toothpaste or chewing gum.
"The idea is to replenish these mucins to the mouth as a protective to boost your ability to protect yourself from caries formation," she explained.
The research makes a fundamental contribution to scientific understanding of host-microbe interactions, Ribbeck noted. "It is generating a paradigm shift from the textbook view of mucus as a simple catchall filter for particles, toward the understanding that mucus is a sophisticated bioactive material with powerful abilities to manipulate microbial behavior."