University of Houston (UH) researchers were awarded $1.4 million to eliminate dental problems that have been linked with buprenorphine, a prescription pain medication that has been shown to reduce opioid cravings, when it’s dissolved in the mouth.
Though it is well known that opioid analogs can affect oral fluids, the researchers will use the funding from the U.S. National Institute of Dental and Craniofacial Research to gain a better understanding of how excessive oral exposure to the medication, which works by changing the way the body responds to pain and can decrease the euphoria experienced with opioids, affects teeth, according to a press release dated December 14 from the university.
Since the drug’s approval in 2002 by the U.S. Food and Drug Administration (FDA) as a medication taken sublingually to treat opioid use disorder, reports of dental problems, including oral infections and tooth decay and loss, have surfaced. The dental issues prompted the FDA to issue a warning about the potential side effects in 2022 and led an Ohio man to file a lawsuit in 2023 against Suboxone drug manufacturers, claiming its use led to his permanent tooth decay.
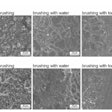
It’s been suggested in the literature that buprenorphine may cause dental problems because it is acidic and the pH of the drug promotes tooth decay. Another proposed cause is that the drug's prolonged contact in the mouth may change the microbial profile of a tooth's surface.
In 2020, an estimated 16 million buprenorphine prescriptions were dispensed from U.S. retail and mail-order pharmacies. An estimated 11 million were dispensed in 2014.
Ming Hu, PhD, a professor at the UH College of Pharmacy, is confident his team can unravel the reasons behind excessive saliva exposure to the drug following sublingual use. Hu's group will use the insights they glean through their research to develop a countermeasure to address these issues.
“It poses a significant concern for individuals with opioid use disorder as the available pharmacotherapy options for this population are very limited,” Hu said in the press release.
When the FDA issued its warning in January 2022, it identified 305 cases of dental problems -- 131 of which were considered serious -- in patients taking buprenorphine medicines that dissolved in the mouth. However, the FDA noted that the number of cases may have been underreported. Later in 2022, the FDA ordered an update to the prescribing information for all buprenorphine products, including Suboxone films and tablets, due to the risk of dental problems.
Along with its warning, the FDA recommended that dentists treating patients taking transmucosal buprenorphine perform a baseline dental exam and a caries risk assessment, establish a cavity preventive plan, and encourage regular checkups.