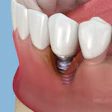
A study comparing the safety and effectiveness of Infuse Bone Graft material to autogenous bone graft when used in maxillary sinus floor augmentation demonstrated that Infuse has similar rates of effectiveness to autogenous bone graft for sinus lifts.
The results were presented at the joint meeting of the American Academy of Oral and Maxillofacial Surgeons and Canadian Academy of Oral and Maxillofacial Surgeons in Toronto and published in the September issue of the Journal of Oral and Maxillofacial Surgery (JOMS; Vol. 67, Issue 9: 1947-1960), according to Medtronic, the company that manufactures Infuse.
The study provided actual human histological evidence that the bone generated by Infuse Bone Graft is normal, mature, 100% viable bone with no residual graft material, as evidenced by core samples taken at the time of dental implant placement.
"Investigators were unanimous in their support of the quantity, safety, and effectiveness of rhBMP-2 in inducing bone. rhBMP-2 is an important alternative to autograft bone for those sinus lift augmentation patients with severe bone loss," said Gilbert Triplett, D.D.S., Ph.D., Oral and Maxillofacial Surgery Chair at the Texas A&M Health Science Center in Dallas and lead author of the JOMS article, said in a press release.
Infuse was cleared by the U.S. Food and Drug Administration in March 2007 for use in certain oral maxillofacial and dental regenerative uses.