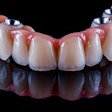
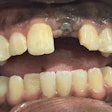
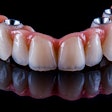
The problem with sintered grafting materials
Conventional bone graft materials on the market today -- whether bovine-derived or synthetic carbonated apatite -- typically undergo sintering, a high-heat manufacturing process that creates dense ceramic particles. While this process is necessary for production or pathogen reduction, it creates significant clinical challenges:
- Prolonged resorption: The dense ceramic structure resists breakdown, often taking up to 12 months to resorb. When normal regenerative pathways are exhausted, particles can become encapsulated, with up to 60% of the grafted area becoming connective tissue rather than bone.1
- Radiographic opacity: Sintered materials often remain highly radiopaque, making it difficult to determine when, or if, material has been replaced by native bone.
- Limited bioactivity: These materials are primarily osteoconductive scaffolds that provide structure but don't actively participate in the regenerative process.
A non-sintered alternative: Bioactive crystal technology
OsteoGen and its family of products are the first and only biomimetic grafting materials featuring proprietary bioactive crystal technology (BCT).
 Dr. Charles D. Schlesinger.Catapult Education.
Dr. Charles D. Schlesinger.Catapult Education.
This unique formulation of nonsintered calcium phosphate crystals combined with type I collagen directly affects the local bone and peri-defect environment on both a cellular and physical level, facilitating bone growth.
Understanding the crystal structure
The foundation of BCT is a nonsintered, highly porous, intertwined calcium phosphate crystal (Fig. 1) with a Ca+ and PO43- ratio similar to natural bone. Structurally biomimetic to laminar bone, it allows bone to grow directly to the crystal surface.2
 Figure 1: Photomicrograph of an OsteoGen bioactive crystal.All images and captions courtesy of OsteoGen.
Figure 1: Photomicrograph of an OsteoGen bioactive crystal.All images and captions courtesy of OsteoGen.
Unlike sintered products, this material is radiolucent upon placement and becomes radiopaque as it is replaced by native bone (Fig. 2), providing clear visual confirmation of graft remodeling and simplifying the timing of implant placement.
 Figure 2: The product is radiolucent at placement and radiopaque upon healing.
Figure 2: The product is radiolucent at placement and radiopaque upon healing.
Clinical evidence: Comparative osteoblast performance
In 2022, Jones et al. demonstrated that osteoblast proliferation on a nonsintered calcium phosphate-collagen matrix was three to six times greater than on allograft and xenograft materials, confirming that denser, sintered materials are less conducive to new bone growth.3
This bioactive approach facilitates bone growth in extraction sites, immediate implant placement gaps, vertical and lateral sinus elevation, and most hard-tissue defects.4
Product forms and clinical application
In products utilizing BCT, the crystals are intertwined in a highly purified type I bovine collagen matrix (Fig. 3). Available in plug, strip, and block configurations, these materials offer flexibility for site-specific grafting applications.
 Figure 3: Composite graft material.
Figure 3: Composite graft material.
In simple alveolar ridge preservation, the plug form is gently packed into the extraction site and secured with sutures. Primary closure is not necessary, nor is a separate membrane or barrier.5 (Fig. 4) The crystal/collagen complex prevents fibroblast penetration and downgrowth while enhancing wound closure compared to other materials.6
 Figure 4: OsteoGen plug sutured in place in a socket preservation.
Figure 4: OsteoGen plug sutured in place in a socket preservation.
Reliable and versatile
In my own practice -- since composite plugs utilizing this technology first came on the market in 2014 and the successive iterations, such as strips and blocks -- I have developed techniques and utilize these products for most of my grafting needs. The overall performance of these bioactive composite grafts in creating quality bone reliably, cost-effectively, and without many of the handling issues associated with other forms of bone graft materials has made them a valuable addition to regenerative dentistry.
For more information, visit Osteogen.com.
References
- Zampara E, Alshammari M, De Bortoli J, et al. A histologic and histomorphometric evaluation of an allograft, xenograft, and alloplast graft for alveolar ridge preservation in humans: A randomized controlled clinical trial. J Oral Implantol. 2022;48(6):541-551.
- Ricci JL, Grew JC, Alexander H. Connective-tissue responses to defined biomaterial surfaces. II. Growth of rat fibroblast and bone marrow cell cultures on microgrooved substrates. J Biomed Mater Res. 1992;26(8):1081-1095.
- Jones K, Williams C, Yuan T, et al. Comparative in vitro study of commercially available products for alveolar ridge preservation. J Periodontol. 2022;93(3):403-411.
- Yosouf K, Heshmeh O, Darwich K. Alveolar ridge preservation utilizing composite (bioceramics/collagen) graft: A cone-beam computed tomography assessment in a randomized split-mouth controlled trial. J Biomed Sci Eng. 2021;14(2):64-73.
- Ferreira CF. Histological evaluation of alveolar ridge preservation using MinerOss® versus OsteoGen® Plug bone graft techniques: clinical study analysis part II. ClinicalTrials.gov Identifier: NCT05419778.
- Kashlan Z, Jafarian J, Fortes Ferreira C, Luepke P. Clinical evaluation of soft tissue closure in alveolar ridge preservation procedures: Part I. J Dent Oral Disord Ther. 2021;9(2):1-8.
Dr. Charles Schlesinger is vice president of clinical development at Impladent Ltd. and an internationally recognized dental implant practitioner and educator. He graduated with honors from The Ohio State University College of Dentistry and completed a general practice residency at the Veterans Affairs Medical Center in San Diego, later serving as chief resident at the Veterans Affairs Medical Center in West Los Angeles. Schlesinger founded the CD Schlesinger Group LLC and was the COO of Comfortable Dentistry 4U. He is a fellow of the International Congress of Oral Implantologists.
The comments and observations expressed herein do not necessarily reflect the opinions of DrBicuspid.com, nor should they be construed as an endorsement or admonishment of any particular idea, vendor, or organization.