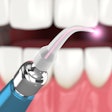
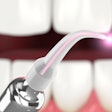
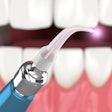
Biolase Technology has received approval to sell its Waterlase dental laser systems in China. The official medical device registration certificate granted by the Chinese State Food and Drug Agency comes after many years of hard work, rigorous testing, and analysis of the product, according to the company.
"Our expansion into mainland China is a very important milestone for Biolase's international development and an initiative we and our partners have been working on for a considerable time," CEO David Mulder said in a press release. "This opens up an exciting new market for us. Dentistry in China is changing very rapidly with the establishment of many new private clinics, and we have already found that Chinese patients are increasingly spending more on modern dental care with innovative dental devices like our Waterlase systems."
Biolase has already developed a strong market presence in Taiwan and Hong Kong through its distribution partners there, Mulder added.
"We have a big head start on our preparations for launching in China, based on our early work in Taiwan and Hong Kong," Mulder said. " We believe the Chinese dental market will expand rapidly with the economic growth in the country, and it is a significant opportunity for us to introduce our dental laser systems at an early stage to what we expect will ultimately become one of Biolase's most important international markets."
Copyright © 2009 DrBicuspid.com