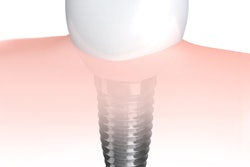
Robot-assisted dental surgery firm Neocis has received U.S. Food and Drug Administration (FDA) 510(k) clearance for a new edentulous indication on its Yomi dental robotic system.
The clearance of a new splint attachment enables Yomi to be used in full arch implant surgeries, as well as in partially edentulous cases, according to the vendor.