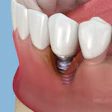
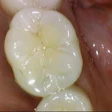
Just when it looked as though the FDA had calmed critics of mercury fillings, the hornet's nest has been stirred up again. Two weeks after the FDA negotiated a settlement in a lawsuit filed by anti-amalgam activists, one of them has moved to reopen the case.
In the settlement, the agency changed the way it describes amalgam on its Web site -- to imply that some patients may be at risk from the substance -- and agreed to reclassify it by July 2009.
Now one of the plaintiffs, Karen Palmer, a former dental assistant, claims she was not notified of the settlement agreement and would not have accepted it if she had been, according to her new attorney, Jim Love, who also serves as counsel for the International Academy of Oral Medicine and Toxicology. She is asking to be removed from the settlement and for the court to consider earlier petitions that call for a complete ban of amalgam.
"There was a mediation scheduled by the court that was attended not by the plaintiffs but plaintiff's counsel, and there was no authority to settle the case on any particular terms," Love said in an interview with DrBicuspid.com.
Charles Brown, the attorney representing the 11 plaintiffs in the original lawsuit against the FDA, says he is shocked by Palmer's sudden opposition to the settlement. He claims she originally agreed to it "until some lawyers started talking to her."
"We didn't anticipate getting a settlement that day, but I was in a settlement room [with the FDA] and we ended up getting the thing we have never had before: an agreement to reclassify," Brown told DrBicuspid.com. "The goal of the case was to get a date to classify and we achieved that, and even more. The FDA has actually gone from praising mercury to advising of its risks. The plaintiffs had a contract to accept reasonable settlement offers, and they were very excited about this settlement agreement. She [Palmer] was very excited about it. I have e-mails from her saying how much she praised the settlement."
But Love maintains that Brown and the FDA had no right to negotiate the settlement agreement without the plaintiffs knowing the details beforehand.
"The settlement was reached by the attorneys for FDA and by Mr. Brown, but you can't limit a plaintiff's or party's right to control the settlement through a fee agreement," he said. "Whether the settlement agreement was reasonable or not isn't the point. The point is that he [Brown] didn't go to Ms. Palmer to get her authority. She was never contacted concerning the terms of the agreement."
In her motion, Palmer says that if she had received notice of the settlement before it was reached, she would not have agreed to its terms. In particular, she "would have agreed to no more than three months for the completion of the classification process," the motion states.
Love says he has already had assurances from FDA counsel that it will go along with the motion to overturn the settlement. FDA spokespeople could not be reached. But Brown is confident that the settlement agreement will move forward as is.
"We will file a motion and we will win that motion," he said. "That settlement will not be overturned because there is no basis for it."