Using neutron imaging, European researchers have shown how the lower mechanical strength in glass ionomer cements results from the presence of pores and also the increased hydrogen mobility within the material, according to a new study in the journal Scientific Reports (March 10, 2015).
"The relationship between microstructure, hydrogen mobility, and strength brings insights into the material's durability, also demonstrating the need and opening the possibility for further research in these dental cements," the authors wrote.
While the placement of fillings is one of the most common dental treatments, a number of these restorations fail within a short period of time, which means general dentists spend practice time replacing failed restorations, they noted.
They estimated that the worldwide costs of these restorations are in the billions of dollars, hence the need for the development of improved restorative materials. Since the signing of the Minamata Convention on Mercury in 2013 by 140 countries, amalgam use is being phased down where possible, meaning these improved restorative materials need to be mercury-free. These alternatives include tooth-colored resins and glass ionomer cements (GICs).
The authors noted that these cements, although not as strong or long-lasting as resin composites, have several advantages that make them suitable a minimally invasive approach, including the following:
- Thermal expansion coefficients similar to the hard dental tissues
- Chemical bonding to enamel and dentine, without the need of an intermediate bonding agent
- Release of fluoride, with consequent positive effects on the re- and demineralization processes involved in the formation of carious lesions
- Biocompatibility
- Antimicrobial properties
"Glass ionomer cement has the advantage that it does not need an intermediate layer of adhesive to bond to the tooth, and it also has the interesting property in that it releases fluoride, which of course helps to prevent cavities. The material also has good biological properties, while it is almost as strong. Our research therefore focuses on understanding the connection between the microstructure of the material and its strength in order to improve its properties," lead author Dr. Ana Benetti, associate professor and researcher at the Odontological Institute at the University of Copenhagen in Denmark, said in a statement.
Imaging
While the microstructure of GICs has usually been assessed by stereomicroscopy or scanning electron microscopy, those techniques are limited to two dimensions, the authors noted. "Accurate information regarding the development of the pore structure following the slow but progressive setting reaction is still missing," they wrote.
A better understanding on the formation of the complex porous structure and the liquid mobility within this structure may help identify possible improvements, according to the authors. The researchers used neutron imaging and x-rays, complemented by biaxial flexural strength, to achieve a better understanding of the microstructure and the photon dynamics of GICs.
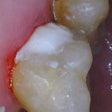
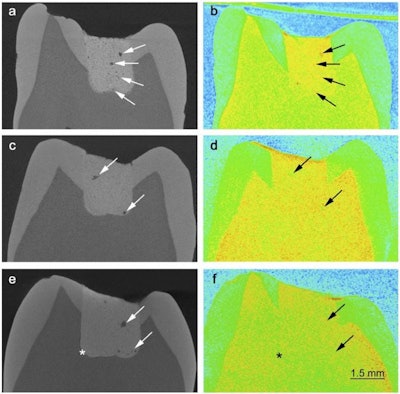 Left: X-rays of teeth with fillings of glass ionomer cement show how porous the cement is. Right: Images of the same teeth using neutron scattering show how the pores are filled with liquid. Credit: Benetti AR et al, DOI:10.1038/srep08972 (2015).
Left: X-rays of teeth with fillings of glass ionomer cement show how porous the cement is. Right: Images of the same teeth using neutron scattering show how the pores are filled with liquid. Credit: Benetti AR et al, DOI:10.1038/srep08972 (2015).The researchers studied two kinds of glass ionomer cement: The cement was the same in both, but a mix of acids was blended into one. They then studied the cement and also the fillings inserted into extracted teeth. The GICs consisted of a powder containing irregular particles of fluoroaluminosilicate glass that undergo an acid-base reaction on contact with the liquid. The GICs were separated into poly and aqua categories. The poly cement (Ionofil Molar AC, Voco) had liquid that contained a polyacid solution, while the aqua cement (Aqua Ionofil Plus, Voco) had water as the liquid, as the polyacid is freeze-dried and incorporated into the powder.
While it is acceptable for the material to be porous, "it is not OK for there to be liquid in the pores, as it can make it easier for the fillings to break," noted co-author Heloisa Bordallo, PhD, associate professor and materials researcher at the Niels Bohr Institute at the University of Copenhagen.
The researchers then took images with neutron scattering and x-ray. By comparing these images, they could now see whether the pores were dry or filled with liquid. To determine, however, how the liquid moved around or attached itself to the materials, the imaging was repeated at separate facilities over the course of days so researchers could observe the chemical reactions that took place.
“You get the strongest material by having cement powder mixed with water that has had acid added to it.”
The less viscous aqua material adapted better to the dental cavity and had a faster setting time, whereas areas of poor contact between the cement and the tooth were identified in the restoration made with the more viscous poly cement, the authors reported.
"Experiments showed that the combination where the acid is mixed up in the cement, so you only have to add water to the cement powder, is the weakest material. You get the strongest material by having cement powder mixed with water that has had acid added to it. So it is better to have the acid in the water -- it helps to bind the liquid faster and stronger to the cement, and there is less water in the pores," Bordallo said.
The authors noted that the "poor adaptation" of the restorative materials within the dental cavity walls and the "network of pores and cracks" in the GIC were concerning. They did find, however, that pore size was reduced in the microstructure over time, backing up previous reports of the increase in mechanical strength of GICs as time progressed.
The study authors reported that they are continuing with research with new cement mixtures containing natural minerals to attempt to reduce the loose liquid in the pores.