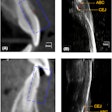
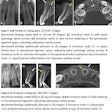
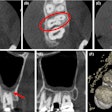
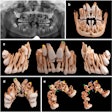
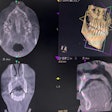
Apteryx has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the 3D module of its XVWeb imaging service.
XVWeb 3D allows dental practices to securely access, view, and share cone-beam CT and stereolithography datasets in real-time while using a compatible web browser.
Apteryx is a subsidiary of dental imaging technology provider LED Medical Diagnostics.